|
230000001629 suppression Effects 0.000 abstract description 7.239000007864 aqueous solution Substances 0.000 abstract description 11.239000000243 solution Substances 0.000 abstract description 28.239000002904 solvent Substances 0.000 claims description 6.XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 10.238000005507 spraying Methods 0.000 claims abstract description 4.239000007884 disintegrant Substances 0.000 claims abstract description 11.230000000996 additive Effects 0.000 claims abstract description 16.239000000654 additive Substances 0.000 claims abstract description 17.239000008187 granular material Substances 0.000 claims abstract description 20.239000011230 binding agent Substances 0.000 claims abstract description 27.230000003179 granulation Effects 0.000 claims abstract description 53.238000005469 granulation Methods 0.000 claims abstract description 53.238000004519 manufacturing process Methods 0.000 title claims abstract description 13.229960004342 cetirizine hydrochloride Drugs 0.000 title claims abstract description 76.ZKLPARSLTMPFCP-UHFFFAOYSA-N levocetirizine Chemical compound C1CN(CCOCC(=O)O)CCN1C(C=1C=CC(Cl)=CC=1)C1=CC=CC=C1 ZKLPARSLTMPFCP-UHFFFAOYSA-N 0.000 title claims abstract description 91.Google has not performed a legal analysis and makes no representation as to the accuracy of the date listed.) Filing date Publication date Application filed by Kyowa Pharmaceutical Industry Co Ltd filed Critical Kyowa Pharmaceutical Industry Co Ltd Priority to JP2006144574A priority Critical patent/JP2007314448A/en Publication of JP2007314448A publication Critical patent/JP2007314448A/en Status Pending legal-status Critical Current Links Original Assignee Kyowa Pharmaceutical Industry Co Ltd Priority date (The priority date is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation or warranty as to the accuracy of the list.) Inventor Tomonaga Aono 知永 青野 Shigenobu Nogawa 重信 野川 Current Assignee (The listed assignees may be inaccurate. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.) Pending Application number JP2006144574A Other languages Japanese ( ja) Google Patents JP2007314448A - Method for producing cetirizine hydrochloride-containing tablet We suggest that cetirizine's reduced incidence of sedative side effects may stem partly from its selectivity for H1 receptors over sites involved in sedation, and partly from its relative exclusion from the CNS.JP2007314448A - Method for producing cetirizine hydrochloride-containing tablet 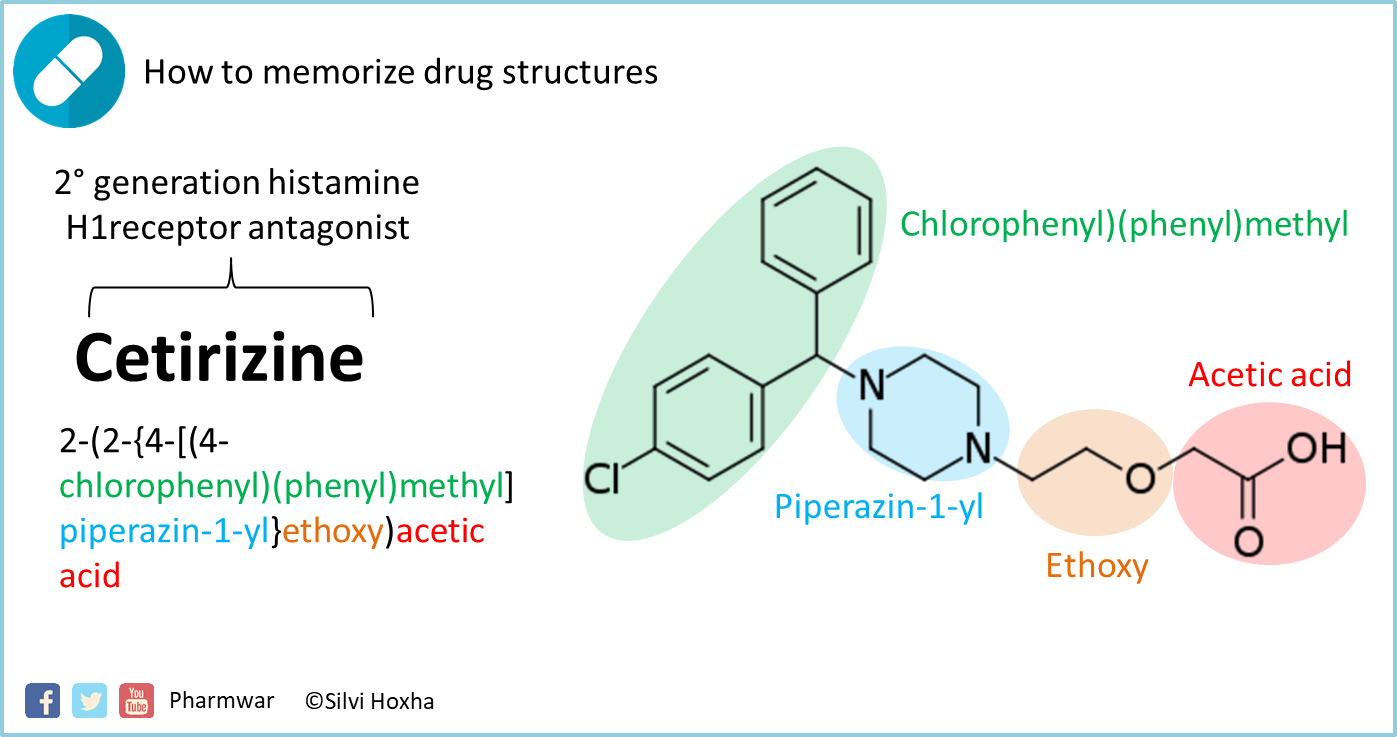
Terfenadine did so to a much lesser extent, and cetirizine passed into the CNS only half as readily as terfenadine. Both hydroxyzine and D-chlorpheniramine crossed the blood-brain barrier in significant amounts. Cetirizine, in fact, failed to bind at any of the receptors investigated except H1 sites, even at concentrations as high as 10 micron. However, their selectivity varied widely. We found the drugs' potency at H1 receptors in the CNS to be similar to their activities in other tissues. We also compared the abilities of cetirizine and three other antihistamines to cross the blood-brain barrier. We compared cetirizine's activity at central H1 sites with the activity of hydroxyzine and terfenadine. This lack of CNS effects may be due to cetirizine's greater selectivity or potency at H1 receptors in the brain, compared with its effects at the receptors involved in sedation, or it may result from the agent's relative exclusion from the CNS compartment. Cetirizine, a carboxylated metabolite of hydroxyzine, possesses the parent compound's antihistaminic activity but does not cause sedation. First-generation H1-antagonist antihistamines such as hydroxyzine have a significant ability to cross the blood-brain barrier and cause sedation, which limits their usefulness in the treatment of allergic disorders.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
